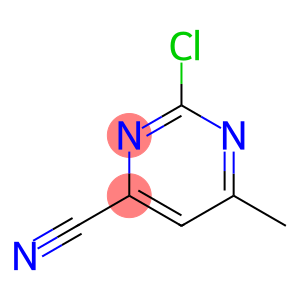
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl-
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl-
CAS: 16858-56-3
Molecular Formula: C6H4ClN3
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- - Names and Identifiers
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- - Physico-chemical Properties
| Molecular Formula | C6H4ClN3 |
| Molar Mass | 153.57 |
| Density | 1.35±0.1 g/cm3(Predicted) |
| Melting Point | 45-46 °C(Solv: ligroine (8032-32-4)) |
| Boling Point | 324.5±22.0 °C(Predicted) |
| pKa | -4.24±0.30(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| UN IDs | 3439 |
| HS Code | 29335990 |
4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- - Introduction
4-pyrimidinecarbonitril, 2-choro-6-methyl-(abbreviated as CMIT) is an organic compound with the chemical formula C5H4ClN3.
CMIT is a white crystalline solid with high solubility. It has good solubility in water and is soluble in a variety of organic solvents such as alcohols, ethers and lipid solvents.
CMIT is mainly used as a powerful bactericide and preservative. It has a broad spectrum of antibacterial activity, can effectively kill a variety of bacteria, fungi and algae. Therefore, CMIT is widely used in industrial and consumer products, such as water treatment agents, paints, paper, cosmetics, plastics and coatings.
The preparation method of CMIT is mainly obtained by the substitution reaction of aromatic amine in the presence of cyanogen bromide. First, an aromatic amine is reacted with phosphonium acid to give the corresponding bromide, and then the bromide is reacted with potassium cyanide to give 4-Pyrimidinecarbonitrile, 2-chloro-6-methyl-.
pay attention to some safety matters when using CMIT. CMIT is irritating to the skin and eyes, so direct contact should be avoided during use. When preparing or applying a CMIT, the operating area needs to be well ventilated and appropriate protective gloves, glasses and respiratory protection equipment should be worn. If you accidentally come into contact with CMIT, you should immediately rinse with plenty of water and seek medical assistance in time.
In addition, CMIT is also an environmental pollutant, and long-term cumulative use may have adverse effects on the environment. Therefore, the use of CMIT should comply with the relevant environmental protection laws and regulations, and properly dispose of waste.
CMIT is a white crystalline solid with high solubility. It has good solubility in water and is soluble in a variety of organic solvents such as alcohols, ethers and lipid solvents.
CMIT is mainly used as a powerful bactericide and preservative. It has a broad spectrum of antibacterial activity, can effectively kill a variety of bacteria, fungi and algae. Therefore, CMIT is widely used in industrial and consumer products, such as water treatment agents, paints, paper, cosmetics, plastics and coatings.
The preparation method of CMIT is mainly obtained by the substitution reaction of aromatic amine in the presence of cyanogen bromide. First, an aromatic amine is reacted with phosphonium acid to give the corresponding bromide, and then the bromide is reacted with potassium cyanide to give 4-Pyrimidinecarbonitrile, 2-chloro-6-methyl-.
pay attention to some safety matters when using CMIT. CMIT is irritating to the skin and eyes, so direct contact should be avoided during use. When preparing or applying a CMIT, the operating area needs to be well ventilated and appropriate protective gloves, glasses and respiratory protection equipment should be worn. If you accidentally come into contact with CMIT, you should immediately rinse with plenty of water and seek medical assistance in time.
In addition, CMIT is also an environmental pollutant, and long-term cumulative use may have adverse effects on the environment. Therefore, the use of CMIT should comply with the relevant environmental protection laws and regulations, and properly dispose of waste.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: 4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- Request for quotation
CAS: 16858-56-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 16858-56-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Visit Supplier Webpage Request for quotationCAS: 16858-56-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Request for quotation
CAS: 16858-56-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 16858-56-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Visit Supplier Webpage Request for quotation
CAS: 16858-56-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 16858-56-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 4-Pyrimidinecarbonitrile, 2-chloro-6-methyl- Request for quotation
CAS: 16858-56-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 16858-56-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Visit Supplier Webpage Request for quotationCAS: 16858-56-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Request for quotation
CAS: 16858-56-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 16858-56-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 2-CHLORO-6-METHYLPYRIMIDINE-4-CARBONITRILE Visit Supplier Webpage Request for quotation
CAS: 16858-56-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 16858-56-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



